


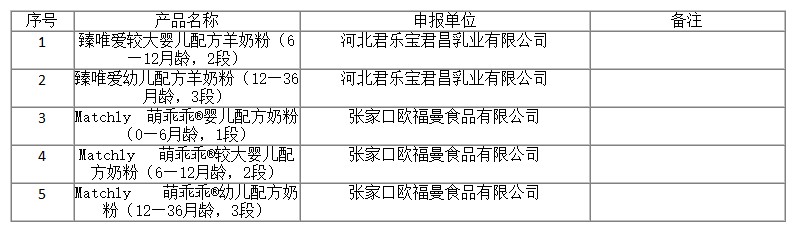
On November 17, 2022, the Center of Food evaluation of the State Administration for Market Regulation updated the list of approval document (decision) for the registration of infant formula product, which includes 5 kinds of infant/older infant/young children formula products, decision details as below: Zhenwei‘ai follow-on formula (6-12 months, stage 2) ,Zhenwei‘ai young children formula (12-36 months, stage 3) , Matchly Mengguaiguai® infant formula (0-6 months, stage 1), Matchly Mengguaiguai® follow-on formula (6-12 months, stage 2), Matchly Mengguaiguai® young children formula(12-36 months, stage 3),etc.
国家市场监督管理总局食品审评中心发布2022年11月22日婴幼儿配方乳粉产品配方注册批件(决定书)邮寄信息,涉及臻唯爱较大婴儿配方羊奶粉(6—12月龄,2段),臻唯爱幼儿配方羊奶粉(12—36月龄,3段),Matchly 萌乖乖®婴儿配方奶粉(0—6月龄,1段),Matchly 萌乖乖®较大婴儿配方奶粉(6—12月龄,2段),Matchly 萌乖乖®幼儿配方奶粉(12—36月龄,3段)等8种婴幼儿配方乳粉类产品。
Need help or have a question?
Send mail